Natural products and secondary metabolites











Vitamin B3:


Dosage:






National Health Service. National Library for Health. http://www.clinicalanswers.nhs.uk/index.cfm?question=248 (Page last accessed on October 31, 2006).
























Natural products and secondary metabolites
Vitamins
Syllabus for Vitamins: clinical aspects of vitamins, and effects of free radicals, synthesis of vitamins: vit-c, nicotinamide, and mechanism of action of vitamins.
Definition:
Vitamins are organic chemical compounds/substances which cannot be synthesized
(in sufficient quantities) in the body but are essential for normal metabolic functions/reactions.
(in sufficient quantities) in the body but are essential for normal metabolic functions/reactions.
Vitamins do not furnish energy and they aren’t used as building units for cellular structures. Lack of specific vitamins leads to distinctive deficiency states such as Beriberi, Rickets, and Scurvy etc.
Classification:
It is convenient for a number of purposes to classify the vitamins as-
- Fat soluble and
- Water soluble.
Fat soluble vitamins:
The vitamins which are soluble in fat solvents (dietary and body fat) but relatively insoluble in water, their absorption from the intestinal tract is associated with that of lipids and are stored in the body in same fashion as fat are known as fat soluble vitamins. Fat soluble vitamins are vitamin A, D, E & K.
It can be noted that fat soluble vitamins might be toxic in large doses and are stored in the body for a long period. Their deficiency state may be caused by conditions that impair fat absorption.
Water soluble vitamins:
These vitamins are soluble in water but generally not soluble in lipid (however some are slightly soluble in certain organic solvents) and therefore easily eliminated from the body.
The water soluble vitamins are vitamin B complex (including vitamin H also known as vitamin B7) and vitamin C. Our discussion will be confined within the
water-soluble vitamins.
water-soluble vitamins.
It should be noted that since water-soluble vitamins are easily excreted, they have greater chance of being deficit than being overabundant.
Vitamin C:
Introduction:
Vitamin C or Ascorbic acid (toxic to viruses, bacteria and some malignant tumor cells) is a naturally occurring vitamin which prevents scurvy and has antioxidant properties. It is a water soluble vitamin.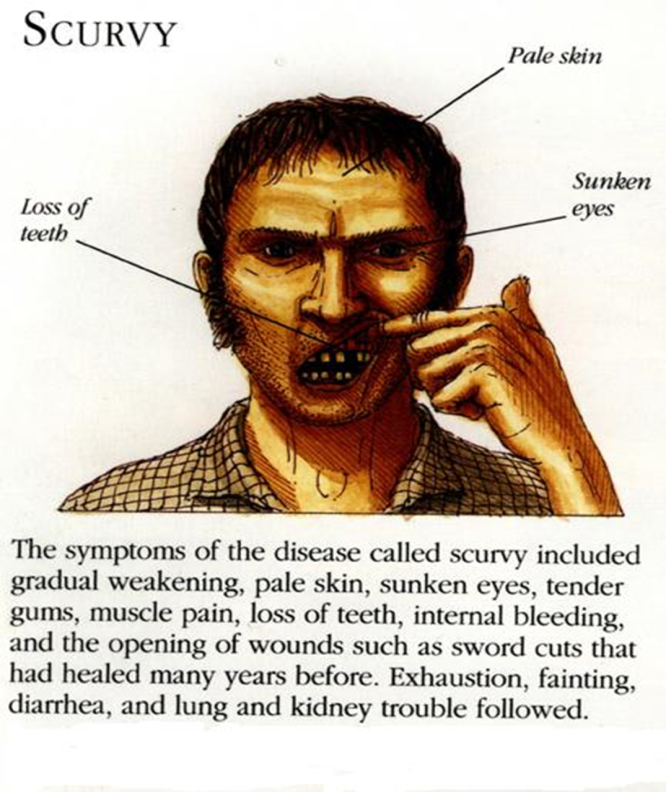
Deficiency symptoms:
Scurvy: It consists of the degeneration of collagen and intracellular ground substances resulting in disturbances of bone growth, hemorrhage of the gums and other part of the body, loosening of the teeth, capillary fragility with consequent cutaneous hemorrhages and other abnormalities.
Synthesis:
The synthesis of vitamin C is done from D-glucose. It is a semi-synthesis.
In this process D-glucose is hydrogenated to D-sorbitol which by oxidation with Acetobactor suboxydans (fermentation) yields L-sorbose. When L-sorbose is treated with acetone in the presence of conc. H2SO4 and the catalyst (used in dehydrogenation) diacetone sorbose is formed. When it is oxidized with KMnO4 in strong alkaline medium diacetone sorburonic acid is formed which upon heating with conc.
HCl gives 2-keto-L-gulonic acid. This when treated with a solution of aqueous HCl in ethanol-chloroform solvent leads to ascorbic acid.
HCl gives 2-keto-L-gulonic acid. This when treated with a solution of aqueous HCl in ethanol-chloroform solvent leads to ascorbic acid.
Uses:
- Prevention and treatment of scurvy
- Facilitation of healing and recovery from extensive burns and severe trauma
- Treatment of some types of anemia (associated with scurvy)
- Maintenance of an acidic urine
Dosage:
- Prophylactic dose → 45-60 mg/per day via oral or IM routes, during pregnancy and lactation an additional 20-40 mg is required.
- Therapeutic dose:
- Treatment of scurvy → 100 mg three times daily for several weeks
- For severe burns → 200-500 mg daily
Mechanism of action
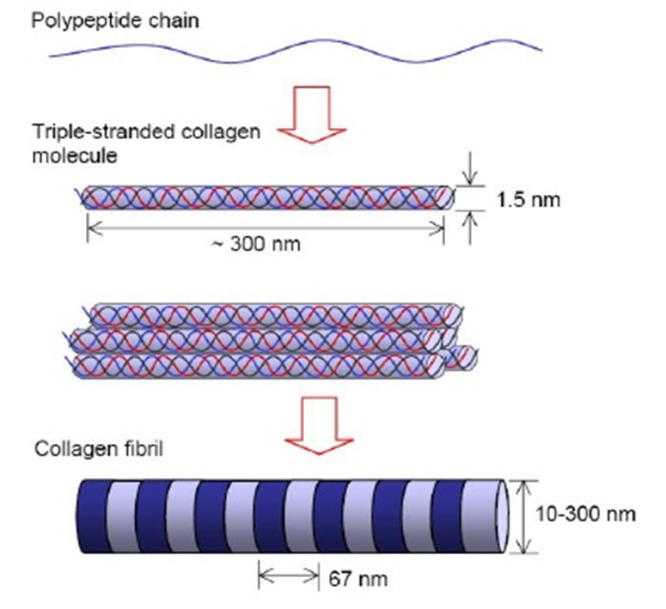
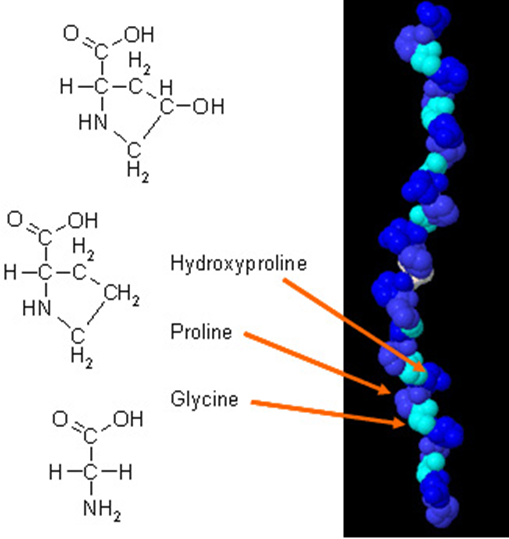
Vitamin C plays a role in many oxidative and other metabolic reactions, e.g. ✓Hydroxylation of proline and lysine residues of protocollagen—essential for formation and stabilization of collagen triple helix;
✓Hydroxylation of carnitine,
✓Conversion of folic acid to folinic acid,
✓Biosynthesis of adrenal steroids, catecholamines, oxytocin and vasopressin and metabolism of cyclic nucleotides & prostaglandins.
✓It directly stimulates collagen synthesis and is very important for maintenance of intercellular connective tissue. A number of ill-defined actions have been ascribed to ascorbic acid in mega doses, but none is proven.
Vitamin C also acts
✓As an antioxidant (reacting directly with aqueous free radicals), which is important in the protection of cellular function; and
✓To enhance the intestinal absorption of nonhaem iron.
Fig. Collagen structure
Vitamin B-complex
B-complex group of vitamins comprise a large number of water soluble vitamins which are nutritional essentials for all forms of life, from the lowest form of yeast and bacteria to the highest form, the man.
Apart from being important nutritionally, they form essential co enzymes to certain important intracellular enzyme systems. There are about individual components, most of them are synthesized by the microbial flora.
Components of vitamin B complex are:-
1. Thiamine – Vit B1
2. Riboflavin – Vit B2
3. Niacin – Vit B3
4. Pantothenic acid – Vit B5
5. Pyridoxine – Vit B6
6. Biotin – Vit B7
7. Folic acid group
8. Cyanocobalamin – Vit B12
Vitamin B1 (thiamine):
Introduction:
Vitamin B1 known as thiamine is a water-soluble vitamin that occurs in moderate to rich quantity in dried yeast, nuts, rice, egg yolks, brans and some other vegetable. It is biotransformed inside the body and function as co-enzyme in several metabolic processes.
Deficiency symptoms:
Beriberi. It is manifested in mainly two forms-
- Dry beriberi → main symptom is polyneuropathy (affect the nervous system)
- Acute wet beriberi → Affect the CVS. Predominant symptoms are edema and serous effusions
- Infantile beriberi →affects the children of malnourished mothers
- Gastrointestinal beriberi→ affects the digestive system and other bodily systems
Synthesis:
Thiamine is rather synthesized than being isolated/ extracted from the food sources. It is synthesized by the direct condensation of 2-methyl-4amino-5-chloromethylpyrimidine hydrochloride and 4-methyl-5-(β-hydroxymethyl) thiazole.
Uses:
- In thiamine deficiency situations e.g. beriberi, neuritis associated with pregnancy and neuritis of pellagra
- Chronic alcoholism
- Wernicke’s encephalopathy
Dosage:
5-10 mg three times daily via oral, IM or IV route.
Biochemical functions (general):
Thiamine functions as a coenzyme in the oxidative decarboxylation of alpha-ketoacids (involved in energy production) and in the transketolase reaction of the pentose phosphate pathway (involved in carbohydrate metabolism).
The enzyme thiamine pyrophosphate (TPP) or co-carboxylase is intimately connected with the energy releasing reactions in the carbohydrate metabolism.
TPP also plays an important role in the transmission of nerve impulse. This is because TPP is required for acetylcholine synthesis and the ion translocation of neural tissue.
Functions of B1 (Specific examples):
1. Enzyme cofactor: (Thiamine pyrophosphate TPP or TDP)
- Decarboxylation reactions
- Pyruvate dehydrogenase
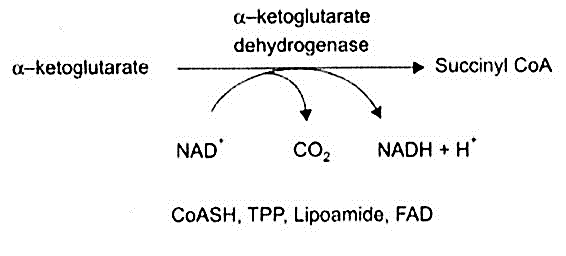
- α-ketoglutarate dehydrogenase
- α-keto acid dehydrogenase – branched chain amino acid metabolim.
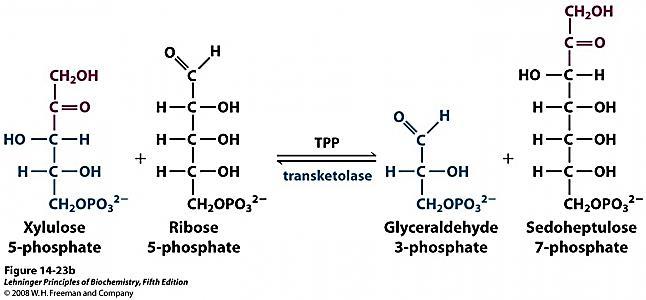
B. Transketolation reactions
- Transketolase – Pentose Phosphate pathway
- Decarboxylation reactions
a) Pyruvate dehydrogenase complexes to TPP:
It catalyzes the breakdown of pyruvate, to acetyl CoA, and carbon dioxide.
b) Alpha ketoglutarate dehydrogenase: 
Requires TPP in the decarboxylation of alpha ketoglutarate to succinyl CoA and CO2.
c) α-keto acid dehydrogenase – branched chain amino acid metabolim.
B. Transketolation reactions
Transketolase: The second group of enzymes that use TPP as co-enzyme are the transketolases, in the Pentose phosphate pathway ( PPP ) of glucose.
Vitamin B2 (Riboflavin):
Introduction:
Riboflavin, also known as vitamin B2 is a
water-soluble vitamin occurring widely in animal and plant foodstuff. It is converted into FAD (flavin adenosine dinucleotide) and necessary for metabolic processes.
water-soluble vitamin occurring widely in animal and plant foodstuff. It is converted into FAD (flavin adenosine dinucleotide) and necessary for metabolic processes.
Deficiency symptoms:
- Well defined deficiency syndrome consists of Cheilosis, glossitis, Seborrheic follicular keratosis of nose and forehead, burning feet etc.
- Loss of hair (alopecia)
- Lesions of the skin, eyes, lips, mouth and genitalia.
Synthesis:
Reaction between 4, 5-dimethyl-N-(1’-ribityl) aniline tetraacetate and 4-nitro-phenyldiazonium chloride yields an azoderivative which upon reaction with barbituric acid yield riboflavin.
Uses:
- In well-defined vitamin B2 deficiency syndrome
- It may also help in deficiency syndromes of other vitamin-B-complex.
Usually after the age of 40-45 vitamin deficiency becomes more pronounced. For example deficiency of vitamin B-complex will lead to lesions of the mouth (and other types of wounds of the mouth). Then vitamin B administration becomes necessary. Usually riboflavin is given with other B-complex vitamins as multivitamin preparations.
Dose:
5-25 mg daily via oral route preferably in a preparation containing other B-complex vitamins.
Functions of B2:
Riboflavin functions as a component of two flavin coenzymes – flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD). Riboflavin through its coenzymes Flavin mononucleotide (FMN) and Flavin adenine dinucleotide (FAD) takes part in a variety of cellular oxidation-reduction reactions and in energy production. Examples include the oxidation of glucose, certain amino acids and fatty acids; reactions with several intermediaries of the Krebs cycle; conversion of pyridoxine to its active coenzyme; and conversion of tryptophan to niacin.
Riboflavin has a role as an antioxidant. It may be involved in maintaining the integrity of erythrocytes.
Some of the specified roles are as follows:
✓Integral component of electron transport chain →ATP Synthesis ----NAD→FMN→CoQ
✓Component of several enzymes in the metabolic pathway
- TCA cycle → succinate dehydrogenase
- Fatty Acid Oxidation → acyl CoA dehydrogenase
- Amino acid oxidation As a part of alpha ketoglutarate
- Isocitrate dehydrogenase complex ( dihydrolipoate dehydrogenase)
☑ FMN-dependent Enzymes
During the amino acid oxidation, FMN is reduced. It is re-oxidized by molecular oxygen to produce hydrogen peroxide.
☑ FAD-dependent enzymes
- Conversion of Succinate to fumarate by succinate dehydrogenase in TCA requires FAD.
- Conversion of Pyruvate to acetyl CoA requires FAD,NAD as part of the Pyruvate dehydrogenase.
- NAD is also required in the a-Ketoglutarate dehydrogenase (Alpha ketoglutarate to succinyl) CoA by alpha in TCA cycle.
Vitamin B3:
Introduction:
Niacin (nicotinic acid) and niacinamide (nicotinamide) are known as vitamin B3.
But niacin show hypocholesterolemic properties not shown by niacinamide and excessive dosage of niacin causes flushing.
Both of these are water soluble vitamins.
Deficiency syndrome:
The well defined deficiency syndrome is pellagra (which is associated with skin lesions, diarrhea and other symptoms). It is characterized by 3D () or 4D symptoms (Diarrhea, Dermatitis, Dementia and Death).
Synthesis:
Esterification of nicotinic acid affords ethyl nicotinate. Amidation of that with ammonia in ethanol results in niacinamide (nicotinamide).
Use:
- Prevention and treatment of pellagra.
Dosage:
For pellagra initially 300-500 mg daily in divided doses preferably in oral route. For maintenance multivitamin preparation is used. In the intravenous route 25-100 mg.
Physiological role and actions
Nicotinic acid is readily converted to its amide which is acomponent of the coenzyme Nicotinamideadenine- dinucleotide (NAD) and its phosphate (NADP) involved in oxidation-reduction reactions.
These pyridine nucleotides act as hydrogen acceptors in the electron transport chain in tissue respiration, glycolysis and fat synthesis. Flavoproteins regenerate them by oxidizing NADH and NADPH.
Nicotinic acid (but not nicotinamide) in large doses is a vasodilator, particularly of cutaneous vessels. It also lowers plasma lipids.
Some of the specified roles are as follows:
Coenzymes are active participants in oxidation-reduction reactions – Dehydrogenases
B3 function in at least 200 reactions in cellular metabolic pathways
NAD+
- Participates in catabolic reactions
- Electron and hydrogen ion acceptor
NADP+
- Anabolic reactions
- Important in biochemical pathway for fatty-acid synthesis, steroid and bile acid synthesis.
NAD+ dependent enzymes
- Lactate dehydrogenase (lactate → pyruvate)
NADPH utilizing reactions
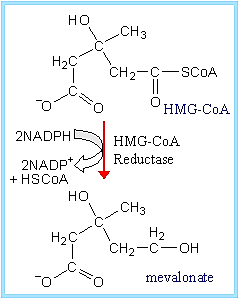
- HMG CoA reductase (HMG CoA → mevalonate) in Fatty acid metabolism
Vitamin B6 (pyridoxine):
Introduction:
Vitamin B6 is actually a mixture of pyridoxine, pyridoxal and pyridoxamine which are interconverted. The most stable and common analogue used in pharmaceutical preparations is pyridoxine.
Deficiency syndrome:
- Seborrheic and desquamative dermatitis of the eyes and mouth
- Glossitis and stomatitis
- Intertrigo of breasts and inguinal region of women
Synthesis:
Uses:
- When using isoniazid or other pyridoxine antagonists causes pyridoxine deficiency. This causes peripheral neuritis. Thus when using pyridoxine antagonists pyridoxine is given as prophylactic.
- Used in other pyridoxine deficiency e.g. convulsions in infants, hypochromic anemia, some types of megaloblastic anemia, homocystinuria etc.
- It improves symptoms related to thiamine, riboflavin and niacin deficiency.
Dose:
- Prophylactic dose → 25-50 mg daily.
- In pyridoxine deficiency syndromes → 10-250 mg daily in oral, IM or IV route
- For drug induced peripheral neuritis → 50-200 mg daily.
Vitamin B5 (pantothenic acid):
Introduction:
Pantothenic acid is a water soluble vitamin widely distributed in nature. It is found in eggs, liver, dairy products and leafy vegetables. It is included in multiple-vitamin preparations and not generally used alone.
Deficiency syndromes:
Deficiency of pantothenic acid leads to several discomforts e.g.
- Malaise (a general sense of discomfort)
- Fatigue
- Headache
- Sleep disturbances
- Nausea
- Abdominal cramps (a sudden and irregular pain)
Dose:
Daily 5-10 mg daily.
Vitamin B7/Vitamin H/Biotin:
Introduction:
Biotin is a water soluble vitamin widely distributed in nature. it is normally synthesized by the intestinal bacteria and therefore supplementation is usually unnecessary.
Deficiency syndromes:
- Mild dermatitis
- Glossitis (inflammation of the tongue)
- Lethargy
- Abdominal pain
- Anorexia
- Mental depressions etc.
Dose:
It is included in multi-vitamin preparations at 0.15 mg/day for children and 0.30 mg/day for adults.
General References
✍ KD Tripathi MD: Essentials of Medical Pharmacology, 7th Edition, Chapter 67: Vitamins, Jaypee Brothers Medical Publishers (P) Ltd, India (2013) pp 909-918
✍ Dietary Supplements, 3rd Edition (Pamela Mason) Pharmaceutical Press, UK, 2007.
✍ Handbook of vitamins, 4th Edition, (Janos Zempleni, Robert B. Rucker, Donald B. McCormick, John W. Suttie) CRC Press,U.S.A. 2007.
Laurence KM, James N, Miller MH, et al. Doubleblind randomised controlled trial of folate treatment before conception to prevent recurrence of neural tube defects. BMJ 1981; 282:
1509–1511.
1509–1511.
National Health Service. National Library for Health. http://www.clinicalanswers.nhs.uk/index.cfm?question=248 (Page last accessed on October 31, 2006).
Alkaloids
Syllabus for Alkaloids: alkaloids as pharmaceutical raw materials, opium and its analogue, synthesis of ephedrine, clinical comparison of ephedrine and epinephrine.
Alkaloids:
Alkaloids may be defined as basic nitrogenous compounds which occur abundantly in the plant kingdom. They contain one or more nitrogen usually in a heterocyclic ring and have a marked physiological action on man & animals. The nitrogen may exist as primary (1° amines), secondary (2° amines) or tertiary (3° amines) amine. These compounds are basic but the degree of their basicity depends on the structure of the molecule and presence and location of other functional groups.
Alkaloids may be defined as basic nitrogenous compounds which occur abundantly in the plant kingdom. They contain one or more nitrogen usually in a heterocyclic ring and have a marked physiological action on man & animals. The nitrogen may exist as primary (1° amines), secondary (2° amines) or tertiary (3° amines) amine. These compounds are basic but the degree of their basicity depends on the structure of the molecule and presence and location of other functional groups.
Alkaloids as pharmaceutical raw materials:
Alkaloids are natural products. They are the secondary metabolites of plant.
Their structure is diverse varying from simple to very complex structure and their activity is also highly selective based on the structure. For example-
Their structure is diverse varying from simple to very complex structure and their activity is also highly selective based on the structure. For example-
The difference between morphine and codeine is that codeine contains a methoxy group in place of phenolic hydroxyl group. Morphine is an analgesic and it is 10 times more potent compare to codeine. But codeine is mainly used as anti-tussive drug. It has mild analgesic.
Again thebaine has structure similar to morphine and codeine (both the hydroxyl groups are replaced by methoxy groups). But thebaine is neither analgesic nor anti-tussive and it is mainly used as CNS stimulant.
Thus we can see that the simple molecular modification of principle compound can give variation of pharmacological action and thus suit the need of different diseases. So it is easily understandable that nature can give us idea of designing drug molecules for different indications.

Opium alkaloids:
Opium:
It is the air dried milky exudates obtained by incising the unriped capsules of poppy plant (Papaver somniferum Linn. of Papaveraceae family).
The plant poppy is an annual herb with large, showy, solitary flowers having color of pink to purple.
Geographical source:
The plant is indigenous to Asia Minor. The plant poppy is cultivated in many countries but legal opium production is not permitted in all countries.
Constituents:
More than 30 (23 according to FA) alkaloids are found in opium. The two principle types of alkaloids present in opium are –
- Alkaloids containing partially hydrogenated phenanthrene nucleus:
Examples include morphine, codeine, thebaine etc which act primarily on the CNS and produce stimulation or depression as well as smooth muscle contraction.
- Alkaloids having 1-benzyl-isoquinoline structure: Example Narcotine, Papaverine. They have little CNS action.
The most important are the alkaloids
- Morphine: It is the most important opium alkaloid. It is a phenanthrene derivative containing an alcoholic and phenolic OH group. Morphine and its salts occur as white silky crystals, odorless and having a bitter taste. They are classified as narcotic analgesics and are strong hypnotic and narcotic. Their use tends to induce habit formation as well as common side-effects as nausea, vomiting and constipation.
- Codeine: It is the most widely used opium alkaloid. It is obtained from opium, or by methylation of morphine or by appropriate reduction and demethylation of thebaine. If methyl group replaces the hydrogen of phenolic OH in morphine it becomes codeine.
The drug and its salts are narcotics and antitussives. They are used as sedatives especially in allaying coughs. Its action is similar to that of morphine but is less toxic.
Heroin: Diacetylmorphine or heroine is formed by acetylation of morphine, where the hydrogen atoms of both the phenolic and alcoholic hydroxyl groups are replaced by acetyl groups. Its action is similar but more pronounced than that of morphine. Due to its potency and the danger of habit formation it is not used in medicine.
- Noscapine
- Thebaine
- Papaverine
Also contains alkaloids like
- Narceine, ii. Protopine, iii. Laudanine, iv. Codamine, iv. Cryptopine,
- Lanthopine , vi. Meconidine
Ephedrine:
Introduction:
Ephedrine is an Alkaloidal amine having sympathomimetic action. Its action is similar to adrenaline but there are some differences.
Ephedrine can be extracted from stems of various species of Ephedra or it can be chemically synthesized.
Chemical synthesis:
- It is synthesized from benzaldehyde by condensing with nitroethane. The corresponding compound in then reduced.
The L-isomer of the reduced compound is then methylated to obtain ephedrine.
- Benzaldehyde is fermented with glucose using yeast. The fermentation of glucose leads to pyruvic acid formation with is condensed with benzaldehyde. The condensed compound is hydrogenated in presence of methylamine to yield ephedrine.
Comparison with epinephrine:
Criterion
|
Ephedrine
|
Epinephrine
|
Route of Administration
|
Effective in oral route
|
Must be given in IV route (except when used as inhaler for asthmatic attack)
|
Duration of action
|
Prolonged
|
Comparatively shorter
|
Onset of action
|
Slower (thus it is more suitable more chronic asthma and colds)
|
Comparatively rapid
|
Potency
|
Comparatively less
|
Greater than ephedrine
|
Blood-Brain-Barrier penetration
|
Greater penetration
|
Penetration is less
|
CNS activity
|
Greater CNS activity
|
Comparatively less active
|
Vasoconstriction
|
Stronger vasoconstriction
|
Comparatively lower vasoconstriction property.
|
Tolerance
|
Possible
|
Usually doesn’t occur
|
Structure
|
Papaverine:
Introduction: Papaverine is a smooth muscle relaxant. It exerts antispasmodic action on blood vessels, thus it relieves arterial spasm caused by acute vascular occlusion. It can be given orally.
Chemical synthesis: Below the synthesis of Papaverine is shown. This is known as Pictet-Gams synthesis.
Cardiac glycosides
Syllabus for Glycosides: chemical and clinical aspects of digoxin and other digitalis glycosides.
Cardiotonics:
Cardiotonics are drugs that acts by increasing the force of contraction of myocardial fiber improve cardiac excitability, automaticity, conduction velocity and refractory period.
In another words, cardiotonics increase the tonicity of the heart i.e. increases the cardiac muscle tone.
Indications of cardiotonics:
- Congestive cardiac failure.
- Atrial fibrillation (The atrial contractions are rapid and irregular. The atrial contraction occurs at a rate of 300-400/minute).
- Atrial flutter (The atrial contractions are rapid but regular. The atrial rate can rise to 250-350/minute).
- Paroxysmal atrial tachycardia (a suddenly occurring arrhythmia where the atrial rate becomes higher – usually 160-200 beats per minute). It is also known as Paroxysmal supraventricular tachycardia.
Effect of cardiotonics:
The cardiotonics increase the force of contraction of myocardial fibers. By doing this a cardiotonic drug –
- Increases cardiac output (the volume of blood pumped out by any ventricle per minute). Increased cardiac output also leads to increased diuresis.
- Lowers venous pressure and venous blood volume.
- In CCF, the pumping is improper and blood volume in heart increases leading to edema in heart. This increases the size of the heart. The cardiotonic counteracts this and decreases the size of heart to normal.
Cardiotonic drugs:
Generally the cardiac glycosides are considered as cardiotonics. There are also some synthetic drugs that may be used for positive inotropic effect.
In acute conditions (acute ventricular failure, tachyarrythmia) synthetic drugs (or ouabain, deslanoside) are used for rapid response. For less acute, chronic or stabilized cardiac failure cardiac glycosides are used. Then, digitalis leaf or digitoxin is 1st choice, digoxin is 2nd choice.
Cardiac glycosides:
Introduction:
Glycosides:
Glycosides are compounds which upon hydrolysis yield a glycone (sugar) part (such as glucose, rhamnose, digitoxose, ribose, cymarose) and an aglycone (also called genin, the non-sugar part) part.
Cardiac glycosides
Cardiac glycosides are glycosides containing a steroidal aglycone and have highly specific and powerful action on cardiac muscle.
They are also called cardio-active glycosides and cardiotonic glycosides. The principle sources of cardiac glycosides are –
- Digitalis
- Strophanthus
- Squill
Digitalis glycosides:
There are about 80 species of Digitalis but only Digitalis purpurea and D. lanata are main sources of cardiac glycosides.
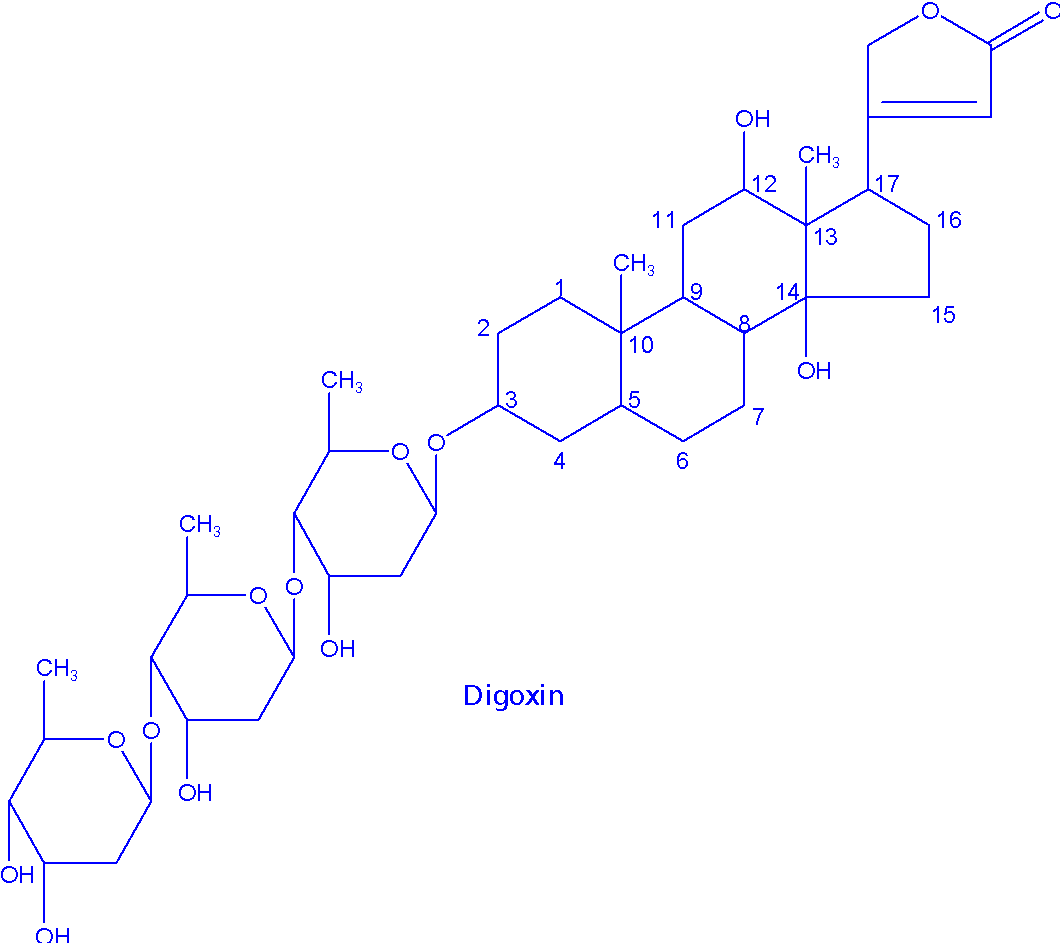
The digitalis leaf refers to dried leaves of Digitalis purpurea. Digitoxin is obtained from these leaves. Digoxin is obtained from dried leaves of D. lanata. Digitoxin and digoxin are the glycosides which are frequently employed as medicine.
Mechanism of action of cardiac glycosides:
General pharmacology:
The definite mechanism of action is not known. Some hypotheses have been postulated –
- The glycosides interfere with the movement of
across the myocardial membrane causing a loss of intracellular
.
- They exert a direct action on contractile proteins in cardiac muscle i.e. actin and myosin.
- They raise the intracellular conc. of
by releasing it from its binding sites and facilitating its entry into the cytosol. It is postulated that they inhibit the
which normally maintains the gradient of the two ions across membranes. Thus when it is inhibited the cell undergoes depolarization. So the ion permeability is changed and
enters the cell. Thus the intracellular
conc. is increased. Then
interacts with actin and myosin leading to contraction of the myocardial fiber. Repolarization reverses this situation.
This theory is most accepted thus far.
Structure of cardiac glycosides:
The following structural features of cardiac glycosides are deemed important –
- Steroidal aglycones known as genins
- Sugars: Attached to the genins in sequence. Most common sugars are D-galactose, D-glucose and L-rhamnose.
- 14 β-OH group.
- 17-α, β-unsaturated lactone ring (6-membered or five membered). If the lactone ring is 5-membered then they are called cardenolides (aka butenolides); if the lactone ring is 6-membered then they are called bufadienolides (aka pentadienolides).
SAR of cardiac glycosides:
The following features are (or were) considered important for pharmacological activity –
- α, β-unsaturated lactone ring at 17-β position.
- Hydroxyl group at 14 –β position.
- cis configuration between the A & B rings and C & D rings.
The following model has been postulated for the binding of cardiac glycoside with the .
.
According to this model –
- First a long range attraction between the lactone ring and site A is initiated.
- Then the steroid part undergoes short-range interaction with site B via hydrophobic bond.
- The sugar unit attached directly with the steroid interacts with the site C which further enhances overall binding.
- This causes receptor to change as follows –
An allosteric effect is thus produced and ATP can’t bind to the protein receptor.
Problems with administration of cardiac glycosides:
- Narrow therapeutic index: The therapeutic index of cardiotonics is very low. Infact it is lowest of all drugs. Their effective dose is 50-60% of the toxic dose. Thus life-threatening poisoning is very common. Such a condition is called “digitalis poisoning.
- Hypakalemia
- Anorexia
- Vomiting
- Salivation
- Diarrhea
- Nausea
Countering problems:
The antidote for digitalis poisoning is Digoxin immune Fab (commercial name Digibind –GSK). It consists of antigen-binding fragments of a specific antidigoxin antibody isolated from immunized sheep.
Other side-effects can be relieved by adjusting the dose.
Commercial preparations of glycosides:
Two drugs digoxin and digitoxin are commercially available for administration.
Digoxin:
Source:
Obtained from the dried leaves of D. lanata (family Scrophulariaceace). It has not been successfully synthesized in laboratory yet.
Use:
It is the most widely used cardiac glycoside for the treatment of congestive heart failure and most supraventricular tachyarrythmias.
It is given in oral or IV route.
Dose:
Dose is individualized. The average loading dose (the initial dose) is 0.75-1.5mg in 1 day when orally given and 0.5-1mg when given in IV route. The maintenance dose (the dose given to maintain the plasma concentration of the drug to therapeutic level) is smaller.
Digitoxin:
Source:
Obtained from the dried leaves of D. purpurea and D. lanata (family Scrophulariaceace).
Use:
It is used in the treatment of congestive heart failure and most supraventricular tachyarrythmias.
Dosage:
Dose is individualized. The general loading dose is 1-1.5mg in 1 day; or 200µg twice daily for 4 days; or 400µg/day for 2-3 days.
Maintenance dose is smaller. Generally 100µg daily or once in two days. The dose may be raised to 200µg/day if necessary.
Get the PDF File & Power point slides